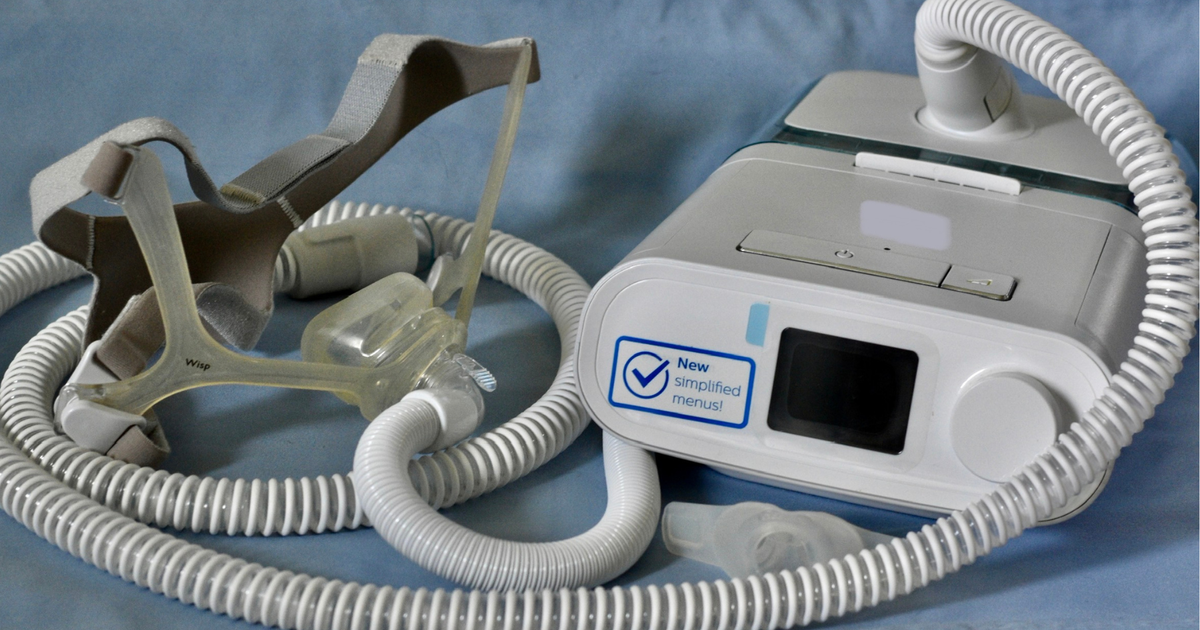
Philips, a company that makes medical devices, has agreed to a special agreement called a “consent decree.” This agreement means they can’t sell their sleep apnea machines in the United States for now. The reason? Some people worried that using these machines could cause health problems, like cancer.
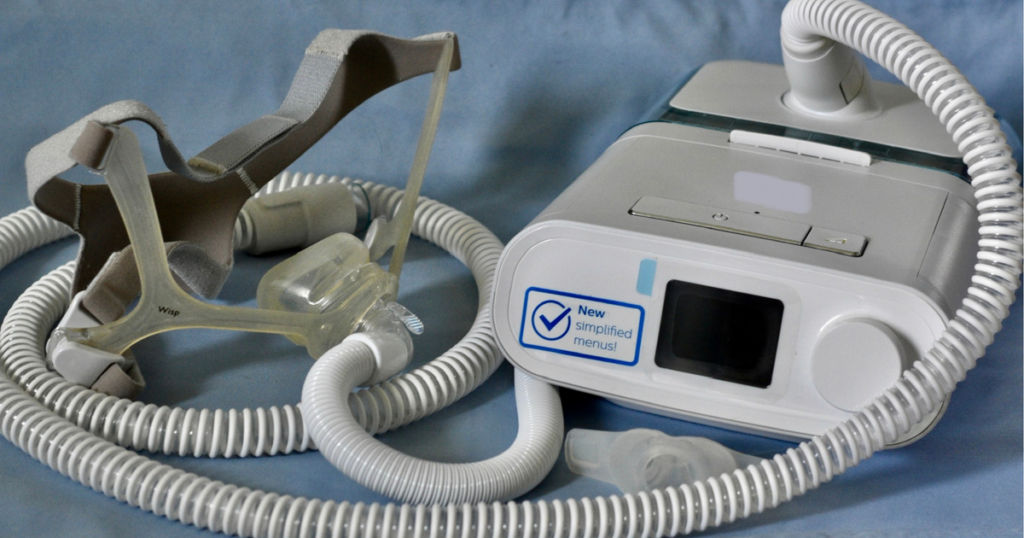
As part of this agreement, Philips has to do a few things. They have to hire another company to check if their machines are safe, do tests at their facilities for five years, and make sure the new machines they send out are okay too. This is the first time a company has to offer a payment plan to help people affected by a machine recall.
If Philips doesn’t follow these rules, they could be fined a lot of money every year. Even though they haven’t admitted they did anything wrong, they agreed to this special agreement back in January.
The head of the U.S. Food and Drug Administration, Jeff Shuren, said they’re doing this to make sure people who use these machines stay safe. The FDA is letting Philips keep making and selling machines that the FDA says are really needed for people’s health.
Philips had to recall millions of their sleep apnea machines in 2021 because of a problem with the foam inside. This foam could break down and make people breathe in stuff while they slept. Although no one has died because of this, it could still make people sick.
The FDA found out that these machines might be connected to over 500 deaths. But Philips says there’s no proof that their machines caused those deaths. However, a news investigation found out that Philips knew about this problem for a long time before telling people.
In 2022, the FDA told Philips they needed to do a better job telling people about the recall and being honest about the risks. But even after that, many people still didn’t know about it.